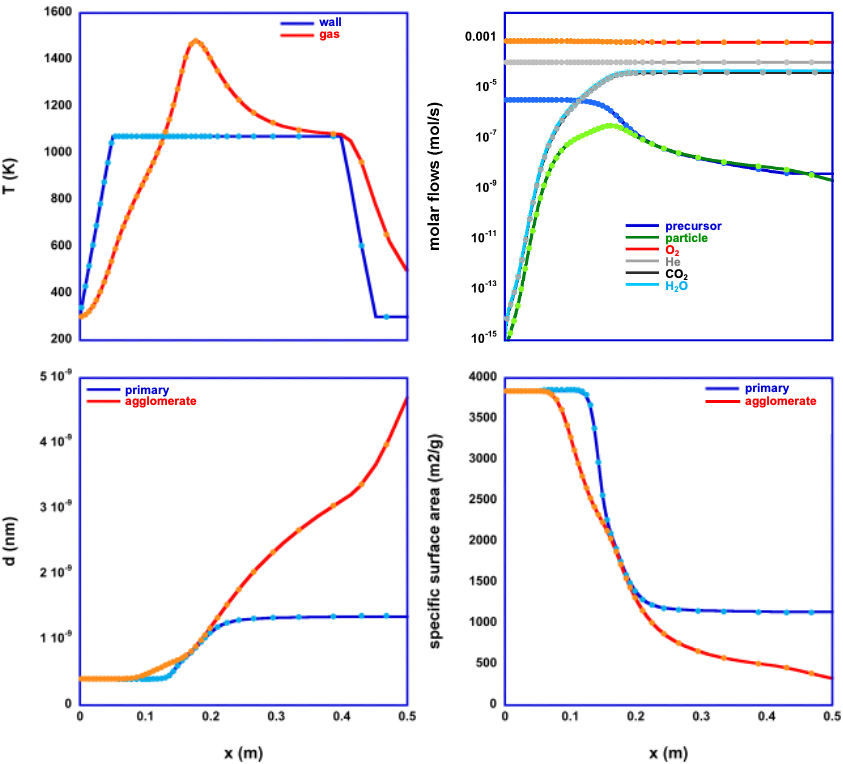
Chemical Vapor Synthesis (CVS) is a method to generate nanoparticles of well defined, small size, low degree of hard agglomeration, high crystallinity and high purity. CVS has relatively high production rates and yields at the lab scale and is scalable.
Several processes act in parallel or in series during CVS of nanoparticles and influence the formation of particles from molecular precursors such as
- conversion of the precursor into monomers (growth species)
- formation of clusters (primary particles or grains) from monomers
- coagulation of primary particles and formation of agglomerates
- sintering of the primary particles in the agglomerates
- heat exchange (with the hot wall) and heat production by the processes 1-4
These processes can be described by a system of coupled differential equations which are numerically solved using a Rosenbrock solver for stiff ordinary differential equations. An important difference between CVD and CVS processes originates in the temperature profile of the CVS (hot wall) reactor which creates an isothermal temperature history for the deposition of films by CVD but a highly non isothermal temperature history for particle formation by CVS, because the position of the substrate in CVD with respect to the reactor temperature is constant while particles have to pass through the complete temperature profile of the gas stream. Therefore, in the program CVSSIN, a simple model for the heat exchange between hot wall and gas as well as the heat production by the particle formation is used.
CVS process parameters designed to optimize nanoparticle characteristics are found using a tool for based on a Monte Carlo type algorithm. This inverse problem of process and materials design is solved for
- primary particle size
- number of primary particles per agglomerate (degree of agglomeration)
- specific surface area (computed from primary particle size)
- specific surface area (computed from agglomerate particle size)
- crystallinity
by variation of - total pressure,
- mass flows and
- wall temperature profile
using a Metropolis type Monte Carlo minimization.
The CVSSIN code is based on the mono disperse coagulation and sintering model of Kruis et al. (Aerosol Sci. Technology. 19 (1993) 514). It was originally developed to describe the synthesis of silicon carbide nanoparticles from tetramethylsilane (Winterer 2002) and has since then been developed to describe the generation of oxides and nitrides (Ognjanovic and Winterer 2018). The code has been extended to design optimized CVS processes using Monte Carlo optimization (Winterer 2018).
The most recent version of the code uses a Rosenbrock instead of an adaptive step size Runge Kutta algorithm to solve the stiff differential equations.
The code is available on request by sending an e-mail to markus.wintererer(at)uni-due.de here: cvssin
- M. Winterer, Discovering paths to optimized nanoparticle characteristics, Chem. Eng. Sci. 186 (2018), 135-141
- S. M. Ognjanovic, M. Winterer, Optimizing particle characteristics of nanocrystalline aluminum nitride, Powder Technol. 326 (2018), 488-497
- R. Djenadic, M. Winterer, Control of nanoparticle agglomeration through variation of the time-temperature profile in chemical vapor synthesis, J. Nanopart. Res. 19 (2017), 1-12
- C. Schilling, and M. Winterer, Preserving Particle Characteristics at Increasing Production Rate of ZnO Nanoparticles by Chemical Vapor Synthesis, Chem. Vap. Dep. 20 (2014), 138-145
- R. Djenadic and M. Winterer, chapter 2, Chemical Vapor Synthess of Nanocrystalline Oxides, in Axel Lorke, Markus Winterer, Roland Schmechel, und Christof Schulz (eds.), Nanoparticles from the Gas Phase – Formation, Structure, Properties, Springer Berlin 2012, ISBN 978-3-642-28546-2
- Markus Winterer, Nanocrystalline Ceramics – Synthesis and Structure, Springer, Heidelberg 2002, Springer Series in Materials Science, Volume 53, ISBN 3-540-43433-X
